 
Consumers should contact their physician or healthcare provider if they have any questions, concerns or have experienced any problems related to using these aerosol sunscreen products. Research suggests regular sunscreen use reduces the risk of potentially deadly. Consumers may also visit for more information and to learn how to receive reimbursement for eligible products. W earing sunscreen every day sounds like a no-brainer piece of health advice. To date, Edgewell has not received any adverse events related to this recall, the FDA added.Ĭonsumers with questions regarding this recall may contact Edgewell Personal Care at 1-88 Monday through Friday, 9:00 a.m. Exposure to benzene can occur by inhalation, orally, and through the skin and it potentially can result in cancers including leukemia and blood cancer of the bone marrow and blood disorders which can be life threatening, according to the FDA. The full list of recalled aerosol sunscreens include: Coppertone Pure & Simple SPF 50, 5oz (Lot TN00CJ4, Lot TN00BR2) Coppertone Pure & Simple Kids SPF 50, 5oz (Lot TN00857, Lot TN00CJV, Lot. No other batches of Hair & Scalp (either before or after these batch codes) and no other Banana Boat products are in included in the recall, the company added.īenzene is classified as a human carcinogen. 6 oz Banana Boat Hair & Scalp Spray SPF 30 Lot Code: 21139AF Expiration Date: April 2024. 
6 oz Banana Boat Hair & Scalp Spray SPF 30 Lot Code: 2008AF Expiration Date: February 2023 Johnson & Johnson conducted a voluntary recall on their spray on sunscreen products (Neutrogena and Aveeno aerosols) and CVS paused sale of two.6 oz Banana Boat Hair & Scalp Spray SPF 30 Lot Code: 20016AF Expiration Date: December 2022 Sunscreen recall 2021: J&J recalling select Neutrogena and Aveeno sunscreen due to presence of benzene Johnson & Johnson is recalling five of.The company said that while benzene is not an ingredient in any Banana Boat products, the review showed "unexpected levels of benzene came from the propellant that sprays the product out of the can." Three batches of Banana Boat Hair & Scalp Sunscreen Spray SPF 30 have been voluntarily recalled by Edgewell Personal Care Company, the parent company of Banana Boat, after an internal review found some samples contained trace levels of benzene. Some Banana Boat sunscreen is being recalled due to the presence of a cancer-causing chemical, according to the Food and Drug Administration. The spray sunscreens affected by the recall are Neutrogena Beach Defense, Neutrogena Cool Dry Sport, Neutrogena Invisible Daily, Neutrogena Ultra Sheer and Aveeno Protect + Refresh. 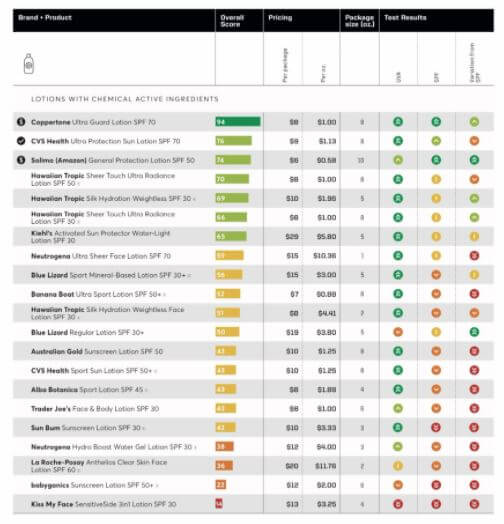
Only the batches above are being recalled.Banana Boat sunscreen was recalled due to the risk of exposure to a cancer-causing chemical
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
